|
1 The Absorb (Abbott Vascular) bioresorbable vascular scaffold (BVS) approval and use has been used as evidence of these phenomena. There has been criticism of the interventional community for having an overly enthusiastic response to new devices and for being slow to abandon them despite safety concerns. New device approvals occur often in interventional cardiology, which is known for its rapid pace of change. These results illustrate an example of an appropriate physician response to adverse data updates and FDA warnings. In addition, declines in use were evident in the subsequent month following the release of data that reported negative outcomes. Declines in use were observed coincident with the scientific presentation of adverse event data as well as FDA warnings.Ĭonclusions and Relevance Most US physicians and hospitals were selective in their use of BVS, primarily using them in patients similar to those in the device’s FDA approval trial. The maximum monthly use of BVS was 1.25% of all PCI procedures that occurred 90 days after FDA approval, but with site-to-site variability. The patient characteristics for BVS use were not dissimilar to the inclusion criteria of the ABSORB III FDA approval trial, with notable differences based on trial eligibility (eg, excluding patients with myocardial infarctions). 
Procedures with implants of BVS occurred among patients with fewer comorbidities and lower-acuity presentations compared with procedures with implants of conventional stents. Of these, 4265 procedures (0.6%) used BVS overall (after FDA approval of BVS). Results Of 682 951 procedures, 471 064 (69.0%) were done in men, 587 301 (86.0%) were among white people, and the mean (SD) age of those undergoing procedures with BVS vs conventional stents was 62.6 (11.4) years vs 65.7 (11.9) years. 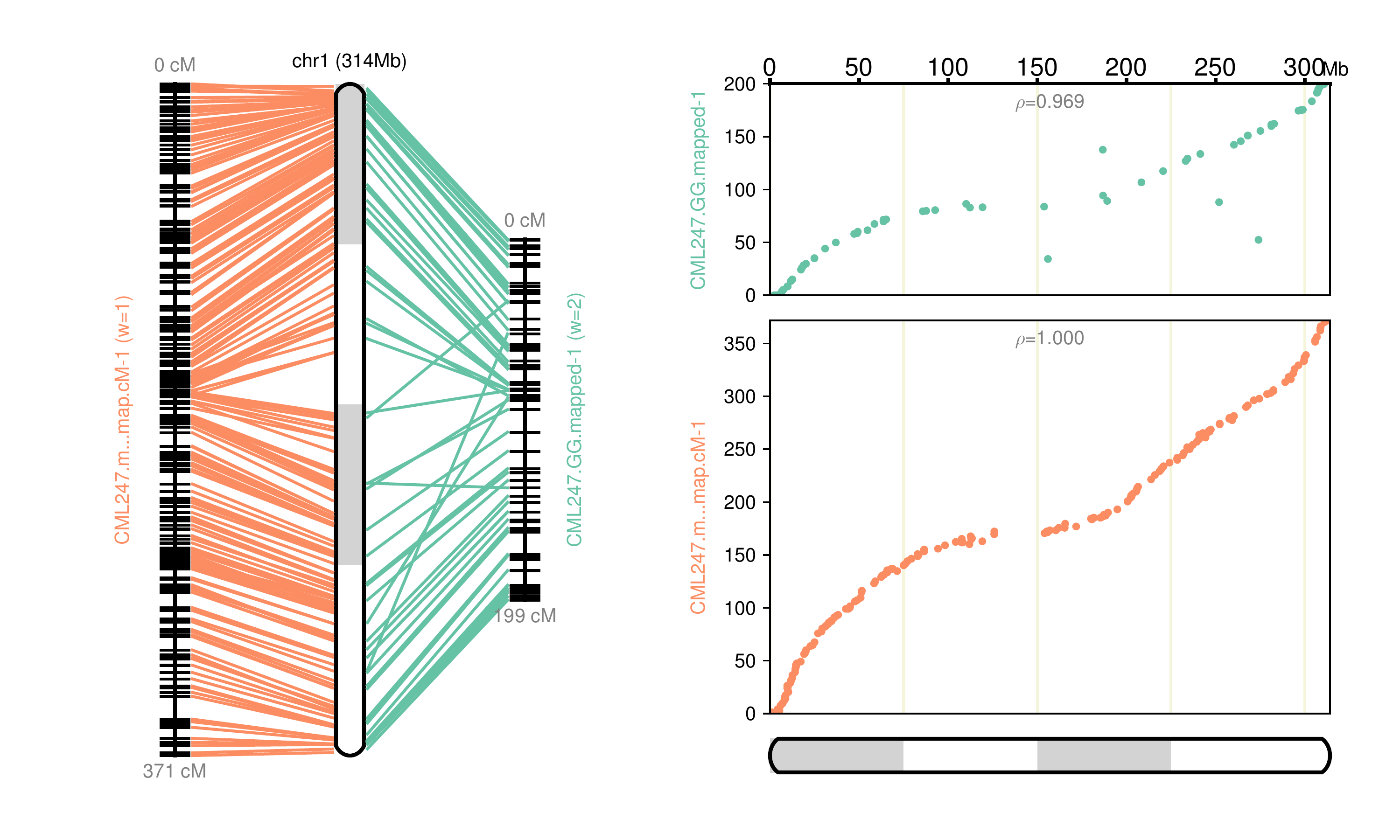
In addition, the characteristics of patients who received BVS and of hospitals that used BVS were assessed and comparisons of patient characteristics between BVS recipients and patients who were treated contemporaneously with metallic stents were made. Main Outcomes and Measures The primary outcome was monthly use of BVS in the United States. Data analysis was performed in October 2017. 
Objective To characterize the patterns of uptake of bioresorbable vascular scaffolds (BVS) within the United States following device approval and to describe changes in response to subsequent releases of data and US Food and Drug Administration (FDA) warnings.ĭesign, Setting, and Participants This analysis of the uptake of BVS between January 2016 and June 2017 used CathPCI Registry data all percutaneous coronary intervention (PCI) procedures with an implant of either a BVS or conventional stent were included. However, to our knowledge, the rates of new product adoption and patterns of new device usage in clinical practice have not been well described. Importance Physicians have been criticized for having an overly enthusiastic response to new device approvals, especially for novel technologies.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
